Digitising your supply chain,
transforming your business
We help Life Science companies design, manage and deliver their packaging, marcoms and digital assets; optimising their digital supply chain
-

Packaging Artwork & Labelling
Write your caption hereButton -

Advisory, Consulting & Project Management
Our consulting team provides strategic advice on supply chain,
packaging/labelling, regulatory, quality, and marketing communications.
Button -

Creative & Digital
Our team delivers best-in-class creativity for the Life Science industry.
Helping you to visually communicate effectively through all channels.
Button -

Strategic Outsourcing – BPS
With a highly competitive and regulated marketplace, it is becoming
the norm within the Life Sciences industry to outsource specialised tasks.
Button -

Software Solutions
Our purpose-built platform, GLAMS, is the world’s only workflow
management system designed specifically for the Life Science industry.
Button
Slide title
Write your caption hereButton
Slide title
Write your caption hereButton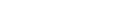
Slide title
Write your caption hereButton
Slide title
Write your caption hereButton
Slide title
Write your caption hereButton
We help Life Science companies design, manage and deliver packaging artwork, marcoms and digital assets
-

Packaging Artwork & Labelling Services
We provide global leadership and best-in-class customer engagement
in the provision of packaging artwork and labelling services for the Life Science industry.
Read more -

Strategic Consultancy
Our consulting team provides strategic advice on supply chain,
packaging/labelling, regulatory, quality, and marketing communications.
Button -

Creative & Digital Services
Our team delivers best-in-class creativity for the Life Science industry.
Helping you to visually communicate effectively through all channels.
Button -

Strategic Outsourcing (BPS)
With a highly competitive and regulated marketplace, it is becoming
the norm within the Life Sciences industry to outsource specialised tasks.
Button -

Managed Services
We manage complex supplier relationships taking full end-to-end
responsibility for the just-in-time delivery that help bring your products to market.
Button -

Software Solutions
Our purpose-built platform, GLAMS, is the world’s only workflow
management system designed specifically for the Life Science industry.
Button
Five solutions, one team and a digitised supply chain
-
Packaging Artwork & Labelling
ButtonLorem ipsum dolor sit amet
-
Advisory, Consulting & Project Management
ButtonLorem ipsum dolor sit amet
-
Creative & Digital
ButtonLorem ipsum dolor sit amet
-
Strategic Outsourcing – BPS
ButtonLorem ipsum dolor sit amet
-
Software Solutions
ButtonLorem ipsum dolor sit amet
We cover five core disciplines with one shared vision: to provide best-in-class supply chain solutions to the Life Science industry. Each of our teams offers expertise and experience across creative, marketing, packaging artwork and labelling, and software – but it’s when they join forces that we’re at our very best. Whether you are using one, two or all of our solutions, you’ll find that our shared knowledge and insight shapes what we deliver, and how.
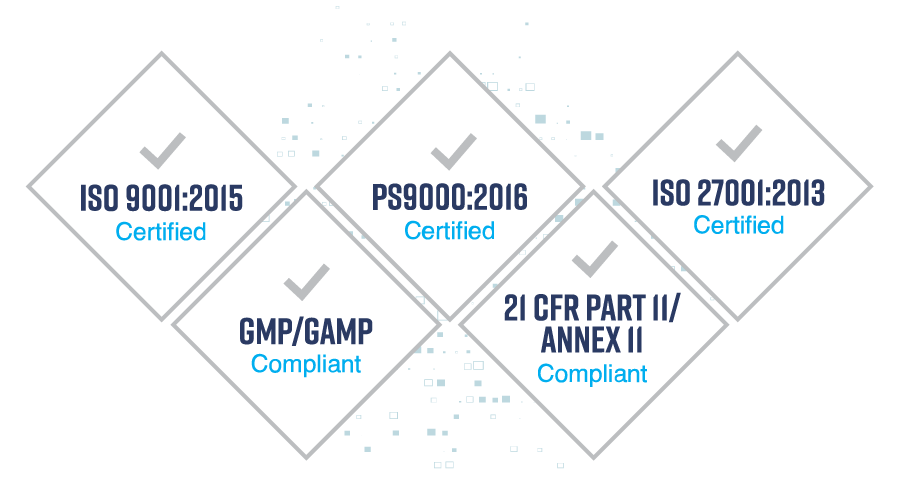
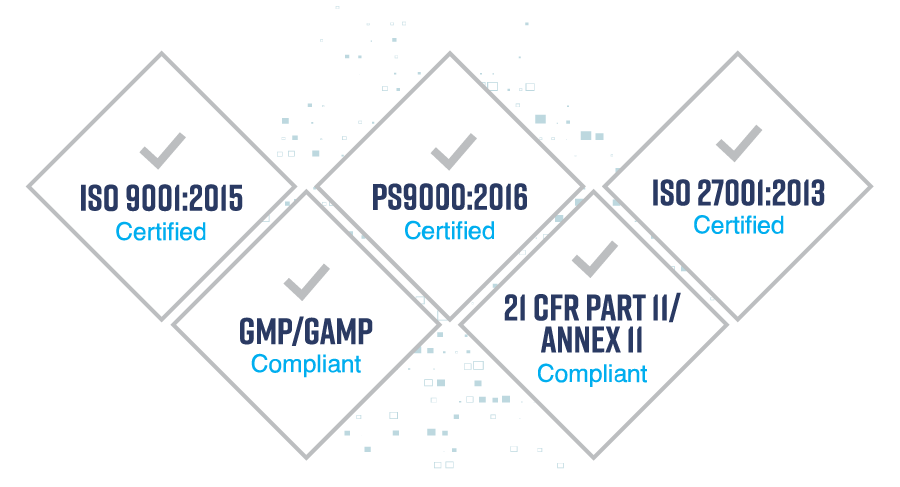
Quality Guaranteed
We enable Life Science companies to digitalise and integrate their supply chain
"Harvesting, sharing rich and live data brings business and customers closer"
Quality Guaranteed
About us
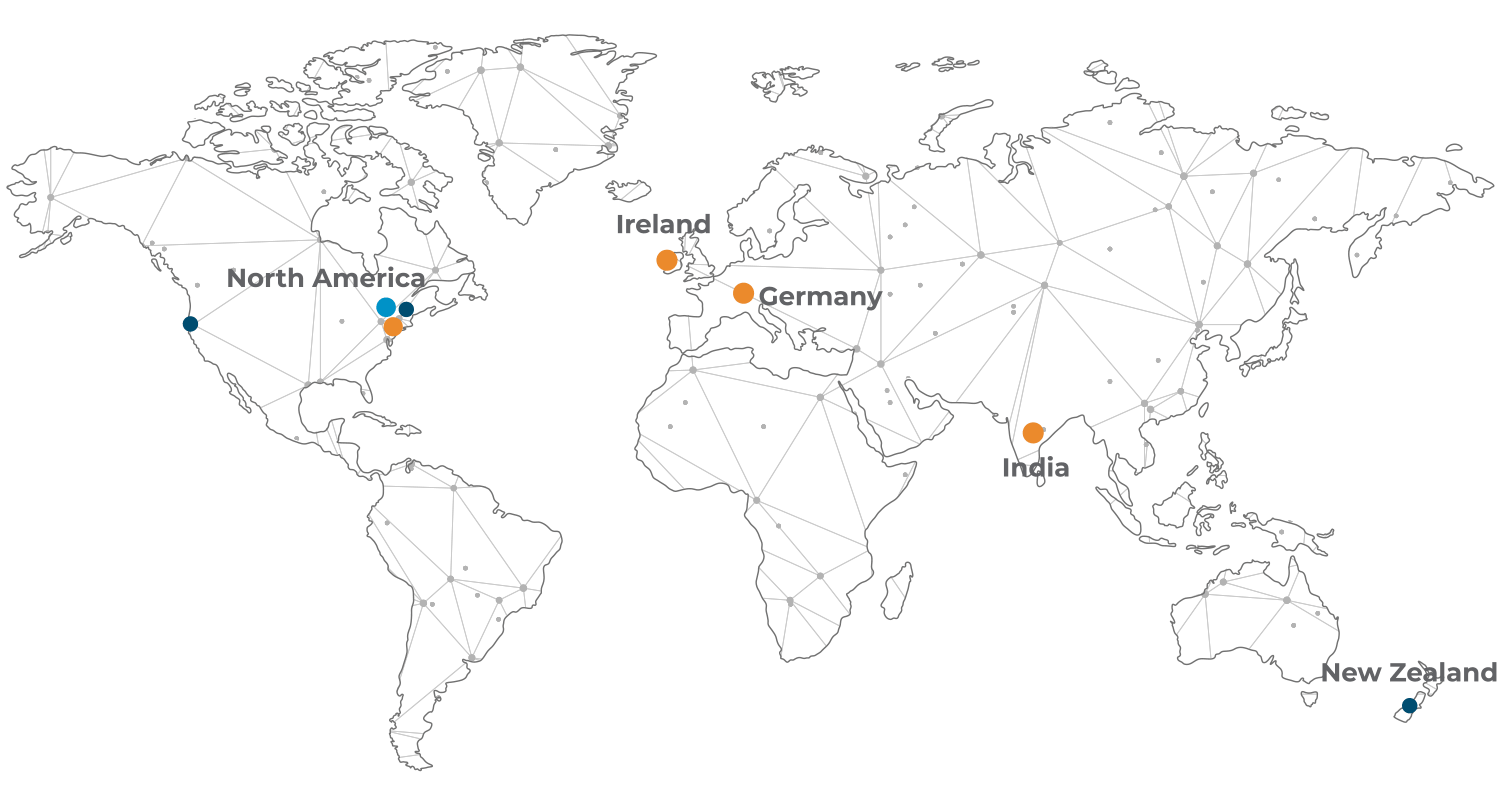
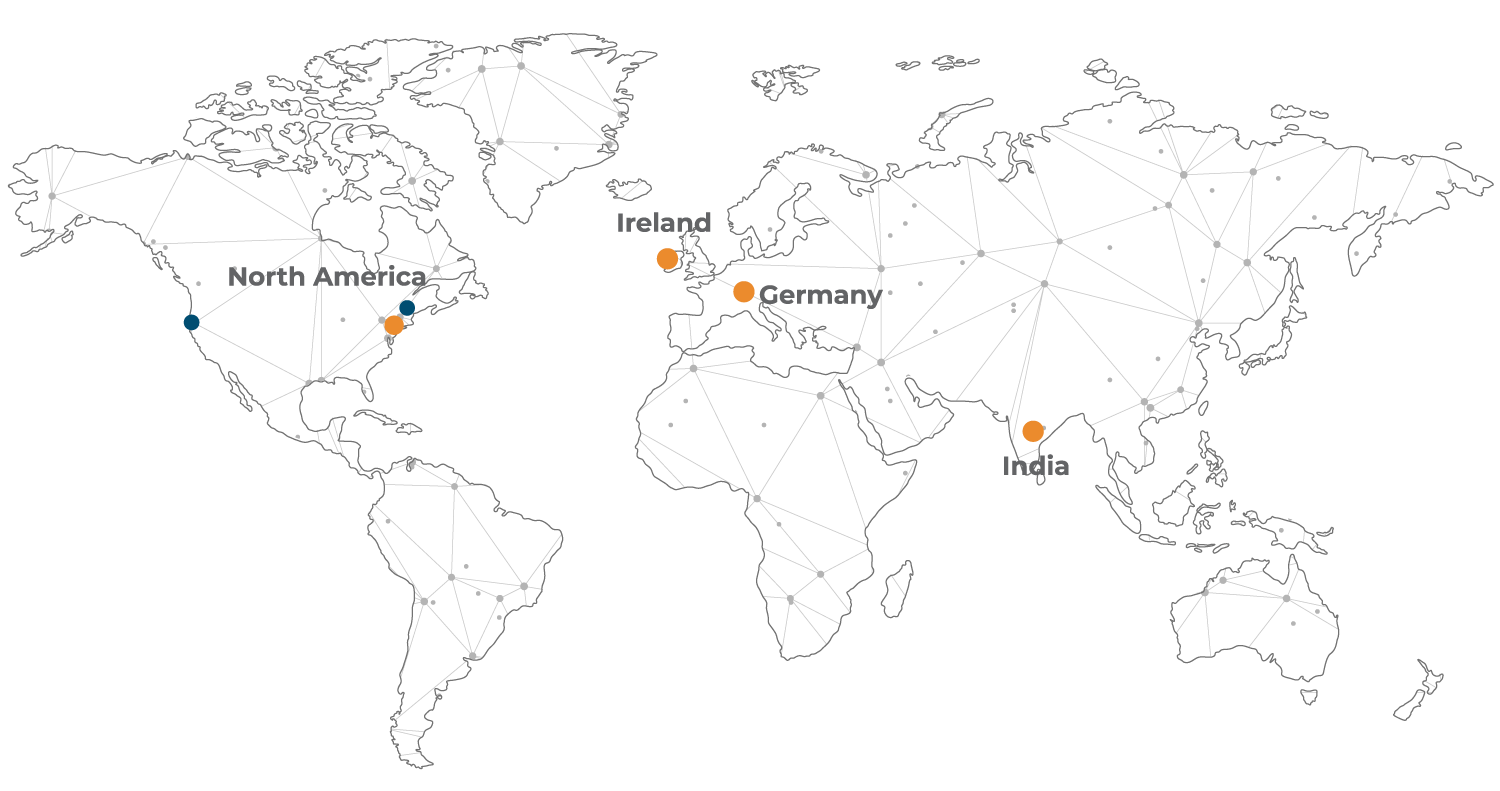
Global Centres of Creative and Packaging Excellence
- Global Centres of Creative and Packaging Excellence
- Dublin, Ireland
- Waldkirch, Germany
- Branford, CT, USA
- Hyderabad, India
- Regional Sales Offices
- Boston, MA, USA
40+
years delivering globally
500+
employees globally
9%
annual R&D spend
100
markets served
Servicing the Life Science industry from our Global Centres of Creative and Packaging Excellence in Europe, Asia and North America, we are committed to helping our clients bring their products to market in the most compliant and creative way possible while at the same time putting patient safety at the heart of everything we do. We deliver our service offering both globally and regionally to best suit our clients market or expansion needs.
We build long-lasting relationships with our customers. The foundation for these relationships and ultimately partnerships is our record of meeting and exceeding our clients’ expectations. We do this through a process of constant analysis, improvement and evolution.
Latest News

Latest News

Events
-

Webinar: Connected Packaging – Enhancing Patient Safety, Medicine Adherence & Brand Awareness
You can now access this Webinar On-Demand
On-Demand Access -

Webinar: Revolutionise your Regulatory Content Change Process
You can now access this Webinar On-Demand
On-Demand Access -
Webinar: Launching your First Drug Product – Key Learnings from Pharmaceutical Start-ups
You can now access this Webinar On-Demand
On-Demand Access -

Join our Perigord Knowledge Centre
To stay up to date with the latest industry trends, artwork innovations and best practice insights
Sign-up Now
Perigord Life Science Solutions A Tech Mahindra Company
Receive Perigord news and updates, and agree to the Privacy Notice by signing-up:
Perigord Life Science Solutions